In Canada, the Standard of Practice for Botox® treatment reflects the expectations that guide how regulated health professionals deliver care. These expectations are shaped by provincial regulatory colleges, clinical evidence, and evolving professional norms.
Rather than existing as a single national document, the standard evolves over time in response to new evidence, quality assurance findings, and changes in care delivery. What is considered appropriate today may differ significantly from past practice.
This shift is especially visible in aesthetic medicine. Botulinum toxin has transitioned from being viewed primarily as a cosmetic service to a medical procedure requiring structured assessment, documentation, informed consent, and risk management.
As early as 2014, training developed by Dr. Roberts at PTIFAT was recognized by a Canadian regulatory college, which engaged PTIFAT to help develop standards for facial injectables. This work has since influenced regulatory guidance across multiple provinces, illustrating how standards evolve through expertise, education, and patient safety priorities. Learn more here.
Key Trends in Evolving Practice
1. Greater Emphasis on Clinical Assessment
Practice has shifted from technique-focused delivery to a structured clinical approach. Clinicians are expected to:
- Conduct and document medical history and contraindications
- Establish a clear clinical indication
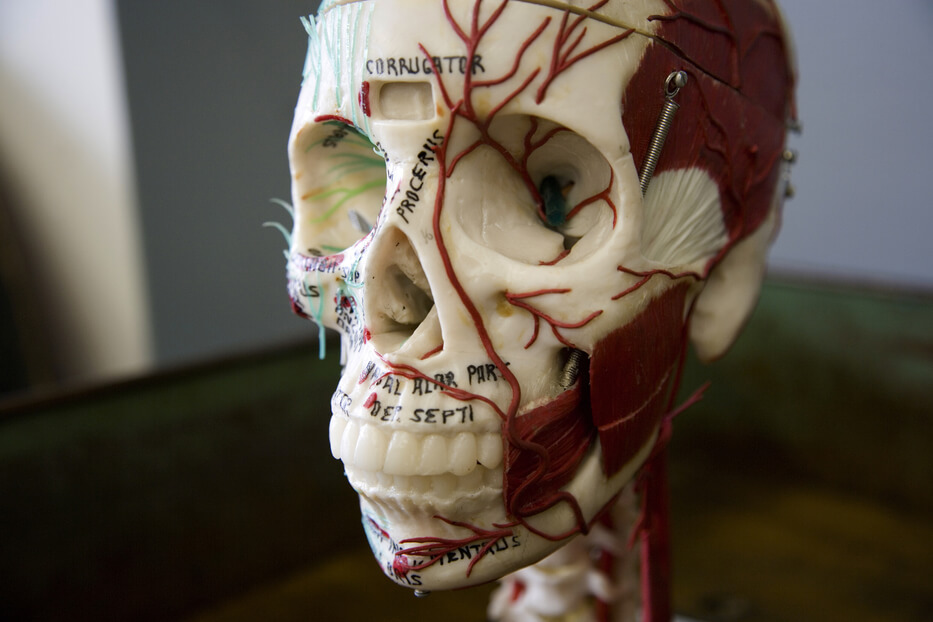
- Align treatment with anatomy, function, and patient goals
This reflects recognition of botulinum toxin as a prescription medication with risks.
2. Documentation as a Core Requirement
Documentation requirements for Botox® treatments have become more rigorous across healthcare, with particularly strong adoption in dental Botox practices, where regulatory scrutiny and structured record-keeping have long been emphasized.
Evolving expectations now include:
- Clear documentation of clinical rationale for Botox® treatment
- Detailed product tracking (dose, dilution, lot number)
- Precise mapping of injection sites
- Standardized before-and-after imaging
Increasingly, clinics are turning to platforms like Facetec to support compliant documentation workflows. Facetec enables structured, secure capture of clinical photography and treatment records, helping practitioners meet rising regulatory expectations.
This level of documentation rigor is now standard in dental Botox, and is rapidly extending into medical, nursing, and naturopathic practices. Across disciplines, regulators are aligning toward a single principle: there can only be one standard of patient care — the highest. As Botox® continues to be recognized as a medical treatment rather than a cosmetic service, consistent documentation practices are becoming essential regardless of provider type.
3. Informed Consent as an Ongoing Process
Consent is no longer viewed as a single form, but as a documented clinical process. It must:
- Be specific to the treatment
- Include risks, benefits, alternatives, and realistic outcomes
- Demonstrate patient understanding
4. Increased Focus on Privacy and Data Security
As Botox® treatments increasingly rely on digital records and clinical photography, expectations around privacy, data security, and patient consent have significantly increased — particularly in dental settings, where compliance frameworks are often more mature.
Clinics are now expected to implement:
- Secure storage of patient records and Botox® treatment images
- Controlled, role-based access to personal health information
- Clear, documented policies for clinical photography and data use
Solutions like Facetec are being adopted to meet these demands, offering secure image capture, consent integration, and privacy-focused storage designed specifically for aesthetic, dental, and medical Botox practices.
This trend, strongly established in dental Botox workflows, is expanding across medical, nursing, and naturopathic professions. Regulators are increasingly applying consistent expectations across all providers performing Botox®, reinforcing that patient privacy and data protection must meet the highest standard—regardless of discipline.
This reflects a broader shift toward risk-based regulation, where clinics proactively adopt best-in-class privacy practices not only for compliance, but to strengthen patient trust and defensibility.
5. Stronger Expectations for Training and Competence
Regulators increasingly emphasize:
- Formal, structured training in injectables and anatomy
- Ongoing professional development
- Practising within scope and demonstrated competence
This reflects the clinical complexity of injectable medicine.
Summary
The Standard of Practice for Botox® in Canada continues to evolve toward a model grounded in clinical reasoning, documentation, informed consent, and data protection. Clinicians who adapt to these expectations not only reduce risk, but contribute to a more credible and patient-centred field. Today, leadership in injectable practice extends beyond technical skill to how care is assessed, documented, and communicated.